With the long dry spells leading into seasons of drought, one’s thoughts turn to the long-held promise that is desalinization. Abutting California, which is facing one of the most severe droughts on record, is the vast Pacific Ocean – if only the salt could be separated from the valuable H2O in an economically feasible way. Enter graphene. The ultra-thin carbon material that has so tantalized the computer industry as a potential semiconductor material might have another use transforming salt water into salt-free potable water. According to new research from Oak Ridge National Laboratory and Rensselaer Polytechnic Institute, a hybrid material, called graphene oxide frameworks, or GOFs, could provide a big advantage over the inefficient desalination processes currently in use.
“This is basically sheets of oxidized graphene connected by specific chemical linkers from some of the oxidation sites,” explains ORNL’s Bobby Sumpter. “Because it’s composed mainly of strongly bonded carbon, it doesn’t decompose in water and has good mechanical properties. It’s an exciting material with potential for numerous applications.”
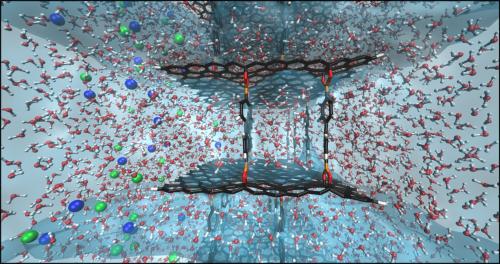
Sumpter and RPI’s Vincent Meunier were initially drawn GOFs’ tunable electronic properties, but with the help of supercomputer simulations performed at RPI’s Center for Computational Innovations soon recognized the material’s potential to be used as a desalination membrane, capable of removing contaminants such as salt ions, pictured below in blue and green, from water.
Among the current techniques for getting salt out of salt water, reverse osmosis systems is used in about 40 percent of desalinzatioplans. The method pushes saltwater through a semi-permeable membrane to generate fresh water, but with reverse osmosis, speed is a limiting factor. The membrane can only handle a certain water pressure.
“You can have a great membrane material but if you can treat only a cup of water a day, that’s not going to be useful or cost-effective,” says Meunier.
Sumpter, Meunier and RPI’s Adrien Nicolaï created computational models at the atomic level and then set out to determine the ideal configuration for a GOF desalination membrane. High-performance computers were used to simulate different elements of the design. The team was concerned with how layer thickness, the density of the linking pillars, and applied pressure all affect the material’s performance.
The sweet spot for removing salt as efficiently as possible involved balancing the selectivity and permeability of the membrane.
The simulations showed where fine-tuning the GOF structure would boost its ability to handle a greater load. It is thought that the new technology can operate approximately 100 times faster than the materials currently used as reverse osmosis membranes. The addition of water-repellent graphene as part of the porous membrane further boosts performance.
“Water is trying to avoid being in contact with graphene, so you can design it in such a way that you’re forcing the water not to be close to one layer but also not to be close to the other,” Meunier said. “This effect creates channels, which direct water through the system very quickly.”
While this research focused on salt ions, the GOF material has other applications, including acting as the filtration membrane for contaminants such as bacteria. Furthermore, GOFs are made with abundant, inexpensive materials through a standard fabrication process, setting the stage for more affordable desalinization efforts.
“We believe it’s scalable, that the chemical engineering industry could potentially produce it in bulk,” Sumpter said.
The results of the research have been published in the journal Physical Chemistry Chemical Physics. Over the last few years the field of materials research has grown by leaps and bounds thanks to computational modeling. The researchers point out how interdiscplinary collaborations and nanoscience are making a big impact in areas that benefit humanity.